Contents
A brief history in garum
There is evidence of fermented fish sauces like garum being consumed as far back as Bronze age (at least 2600 years ago), but because nothing was recorded then, the details are very scarce. As you move into the Iron Age, there is an increasing amount of evidence that fish sauces are an important part of life in Europe, particularly in the Celtic regions. In fact, the most Western part of France which was known as Armorica was renowned for producing a high quality garum which would ‘give you strength‘ (Interestingly, the fictional characters Astrix and Obelisk who fought the Roman Empire lived in this region. Perhaps the secret potion that gave them super-human strength was garum?).
Legends aside, fermented fish sauces were certainly consumed in Europe before the Roman Empire expanded into France and Britain, but it was the Romans who made it popular across the continent, and made it commercially.

Evidence of garum factories are found throughout the Roman Empire, and from these factories it is clear that they made 2 types. One was plain garum, which was made in vast quantities and was used regularly as a flavour enhancer for food. The other was made in much smaller quantities for medical purposes and was called garum armoricum (after the region in France). A popular fish used to make high quality garum armoricum was the tunny (or tuna), which could only be caught once a year during their migration to spawning grounds. The limited amount of tuna available year round combined with the high quality garum it produced made it a very expensive and sought after product. As tuna is a very oily fish the resulting garum would likely be high in omega-3 fats, which may well be one reason it was associated with promoting health.
This garum was used by the military, athletes and women/ children considered to be ‘weak’, because it was associated with promoting strength and good health. It is clear that its benefits were well recognised throughout the Roman Empire. However, as the Roman Empire went into decline the price of salt (which was essential for garum) rapidly increased which meant that garum could no longer be made. After this point in Europe fish sauces were never made on the same scale and were largely forgotten about, aside from some small isolated communities.
Production
Recipes for garum vary from region to region, but they all follow a basic blueprint. Small fish (or the organs of specific fish such as tuna) are layered in containers. Between each layer of fish/ fish organs is a layer of salt and sometimes herbs. This is then left to ferment (often in the sun) for 1-6 months. The liquid can then be removed and shipped all over the empire as a flavour enhancer or medicine.
Using the same basic recipe that the Romans did, Green Pastures created a garum-esque cod liver oil. Green Pastures mix cod livers with salt and a starter broth, then let it ferment for a number of months before the oil is filtered out. From speaking to David Wetzel of Green Pastures, the only real difference between what the Romans did and what they do is the use of a starter broth. Green Pastures save some of the brine from previous ferments to use for future production. This ensures that the same bacteria are doing the fermentation, and so keeps the end product consistent (similar to how cheeses are made). Aside from this, Green Pastures ferment their cod livers in the similar way the Romans did.
Production is simple and traditional, but to anyone familiar with lipid biochemistry this method doesn’t sound like it would produce a healthy oil. Fats in these fish are predominantly poly-unsaturated fats, which are extremely easily oxidised. Being left in these conditions for an extended period of time seems like it oxidation is inevitable, which would of course make these fats toxic.

Roman fish salting pits (garum production)
Stability
The Romans and Gauls didn’t know the details of what was going on in this mix, all they knew was that garum was good for you. However, with modern research we can look much more closely at what is going on in production of garum.
Microbial stability
The high salt content is extremely important because it controls which bacteria ferment the product. The process of fermentation is very similar to that of rotting, but with a key difference. Fermentation is controlled, whereas rotting is uncontrolled. This means that in a fermented product you control the type of micro-organism involved, the conditions of the fermentation, and the duration, which allows you to dictate what the final product is. Rotting on the other hand is uncontrolled, and so the end product is always unknown (and often bad for you).
The very high salt content prevents food borne pathogenic bacteria growing1, yet is tolerated by specific species of bacteria including Lactobacillis spp and Enterobacteria spp2,3. Here they thrive and produce lactic acid as a bi-product of the metabolism of carbohydrates (from either glycogen in the fish organs or in added herbs). The lactic acid lowers the pH of the mix, which further helps to prevent other species of bacteria colonising the product. This makes the fermentation very stable, and makes it near impossible for other species of bacteria to colonise. This is a similar process to fermenting other foods including fermented sausages and fermented vegetables.
Chemical stability
The chemical stability of the product is of the utmost importance because poly-unsaturated fats are extremely vulnerable to oxidative damage. There is also a relativity high amount of free fatty acids present in the oil (up to 20%) which are susceptible to oxidative damage. Some fatty acids are naturally occurring, but others are a result of hydrolysis of the tryglycerides by the fermenting bacteria. Hydrolysis is a process which breaks the fatty acids from their glycerol backbone with lipase enzymes (the same way our digestive system breaks down fats) to release the fatty acid molecule. These broken down triglycerides pose no risk in themselves, and are quite natural (you could even argue that they would be easier to absorb). However, their presence along with the high poly-unsaturated fat content does make the risk of oxidative rancidity a concern.
Despite the concerns, we know that the oil is stable (discussed below), but how the oil remains so stable isn’t fully understood. There are a number of factors which could explain or at least contribute to the stability, but there is certainly more research needed to know the details:
Anaerobic conditions – Lactobacillius spp are a type of bacteria called facultative anaerobes, which means they can use oxygen when it is present, but are able to survive without it. During fermentation these bacteria will quickly metabolise any oxygen present in the mix and then switch to anaerobic respiration. This produces carbon dioxide which will bubble to the surface and form a layer of carbon dioxide, which prevents oxygen reaching the oil. Without any oxygen able to reach the oil, the risk of oxidation is dramatically reduced.
Carotenoids – The fish themselves have highly refined and effective antioxidant systems which are needed to stop their high fat content oxidising and poisoning themselves. Fish and many types of seafood contain a variety of carotenoids (such as astaxanthin). There has been over 180 different types of carotenoids found in marine life so far5, and there are sure to be more identified. Some can to be converted into vitamin A, but many cannot, and their role in fish isn’t fully understood. However, research has shown that virtually all carotenoids are powerful antioxidants and are able to protect lipids from oxidative damage very effectively. Some even have self regenerative properties which allows 1 molecule to quench 1000 singlet oxygen molecules. They certainly have some impressive protective properties, but their abundance in the oil combined with the lack of understanding makes it difficult to understand the extent of their protection.
Quinones – Fish tissue is very high in various quinones, such as Co-Q10, which are known fat soluble antioxidants. How they act as antioxidants is still conjectural – they may recycle other antioxidants (such as carotenes) or act as primary antioxidants themselves. We do know that they exert impressive antioxidant properties, but again, with so little known about their abundance in the oil and their mechanics and impact hard to quantify.
Amines, amino acids and peptides – all of these have a complicated potential to exert strong antioxidant activity. Amines are of particular interest because although they are naturally present in the fish, some of the fermenting bacteria (such as the enterobacteria) can produce amines from the fish proteins6, which increases their concentration. Amines, amino acids and peptides can all act as primary antioxidants, bind with pro-antioxidant metals or synergyse with other antioxidants. This provides a very diverse and flexible form of antioxidant activity. In particular, the amines putrescine, spermidine, and spermine are known to have lipid protective properties7, and these have been identified in the oil produced by fermenting cod livers. Again, the extent of the impact on the oil isn’t really known, and although we know various amines are present in the oil, we do not know what the content of amino acids is, nor do we know if the antioxidative peptides are even present.
Vitamins – Vitamin A, which is abundant in marine oils is a known antioxidant. It is chemically similar to the carotenoids previously mentioned, but its mechanisms are much better understood. Vitamin A is known to have an important role in preventing lipid peroxidation8, and so would naturally protect the fats in this product. Marine oils are also abundant in vitamin D, which protects against lipid peroxidation through different mechanisms to vitamin A – offering another dimension to the protection. In addition to this, other vitamins such as vitamin E are found in marine oils (although to a lesser extent than vitamin A and D) which are also known to help protect lipids.
Other – There are a whole host of other chemicals in fish oils, the activity of which isn’t understood. Chlorophyll for example is found in many fish oils (before filtration), which is a known antioxidant and could support the stability of the oil. In addition to this, there may well be other systems in place that we haven’t even identified. The bacteria themselves can produce all kinds of chemicals and have their own antioxidant systems in place to protect themselves. These could very well help to protect the oil against radical damage
You can see that there is a lot of potential for creating a very powerful antioxidant environment which will protect the oil. What we cannot say for certain is just what is responsible for the stability, and there is every chance that the chemical stability may not even be due to the compounds mentioned above.
Evidence of stability
With so little known about the antioxidant systems involved in the oil, it is easy to be sceptical that they even exist at all. I’ve said we know the oil is stable, but how do we know this?
Understanding the ‘how’ of stability is quite complex, but understanding the ‘if’ is relatively simple. We know that the oil contains an abundance of poly-unsaturated fats and free fatty acids, and we know that in an unstable environment these fats oxidise and undergo a series of chemical changes:
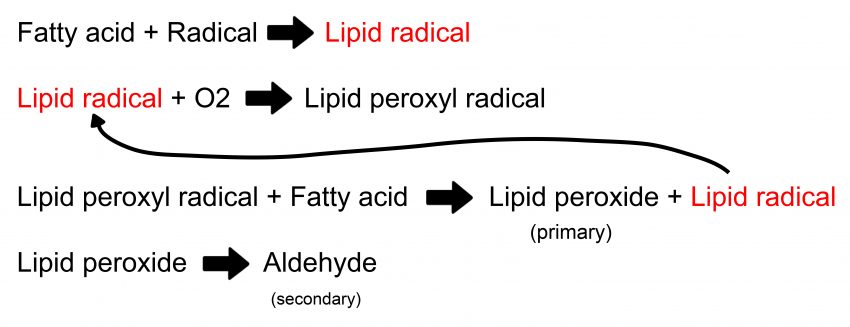
The above image illustrates the stages an oil undergoes when it experiences oxidative rancidity. From the image you can see that the lipid radical created in the 3rd stage can be recycled to initiate the 2nd stage. This causes a chain reaction which can rapidly degrade fats into aldehydes (a common one being malondialdehyde9). The aldehydes themselves are not particularly stable, and they can eventually break down further into smaller volatile compounds. However, what is important here is when the oil isn’t stable, the fats will be converted into lipid peroxides and aldehydes. This chemical change can be very easily tested in a lab.
One simple method is to test for the presence of fats in the oil (the presence of them shows that they haven’t degraded). Below are some lab tests from Green Pastures oil, and highlighted is the percentage of each type of fat. Combined, they account for 99% of the oil – showing fats haven’t been chemically changed.

You can double check this, and you can test for aldehydes. The common test for this is a TBA test which binds to malondialdehyde (a commonly created aldehyde when polyunsaturated fats become rancid)10. The results of this test can be seen below, and have come back very low.
![]()
It is worth noting that there are some limitations to the TBA value test, and it can give abnormally low readings in some circumstances. However, the lack of malondialdehyde is a very promising sign. (There are other similar tests you can do for breakdown products, but they all have their equal share of short-comings, and at any rate, the ones conducted on fermented cod liver oils have had very low values).
You can also triple check – have a look at the fats present. Certain fats are more vulnerable to oxidation than others. Long chain poly-unsaturated fats are some of the most vulnerable, and looking for the presence of these will indicate how reactive the environment is. If an oil is reactive, you typically wouldn’t expect to see any of these because they would be the first to break down.
A good poly-unsaturated fat to look at would be the omega-3 fatty acid DHA. It has a long 22 carbon chain with 6 double bonds, which are the sight of oxidative damage. You would also expect a substantial amount to be found in the oil produced from cod livers. Back to the lab results:
![]()
DHA makes up 7.43% of the oil, which is within the levels you would expect in this type of oil.
From the test data, it is clear that the fatty acids are not damaged. Although this gets us no closer to how, it does show that the fats are protected.
What makes this oil all the more mysterious is that aldehydes cannot be created in this oil using methods that would in other marine oils. You can quite easily force the oxidation reaction with various heat experiments, but for some reason, you can’t in this oil. This raises far more questions than it answers, and illustrates just how little is known about this oil. Nevertheless, it is an interesting observation which I feel is worth adding to this discussion.
Summary
Marine oil ‘supplements’ like cod liver oil are not as much of a new invention as most people would expect. Evidence of people enjoying the oily juices of fish like tuna as a medicine go beyond the Roman Empire. Back then, just like today, their health benefits are well recognised (although not understood). The processing methods used to make products like garum were very safe, and chemically stable. Green Pastures re-created the same production methods that the Romans used to create a garum-like cod liver oil. When testing the oil they found it to be very chemically stable. Despite all the modern analytical techniques, we still cannot say what is making this oil stable – all we can do speculate and identify potential mechanisms.
References
1. Microbial Ecology of Foods, 1980, Vol.1, Chapter 4
2.Anita MENCONI. (2014). Identification and Characterization of Lactic Acid Bacteria in a Commercial Probiotic Culture. Biosci Microbiota Food Health. 33 (1), 25-30.
3. F. Tabatabaei-Yazdi et al. (2103). Effect of temperature and salt concentration on microbial changes during Tarkhineh fermentation. Scientific Journal of Biological Sciences. 2 (1), 8-16.
4. MidWest Laboritories. (2015). REPORT OF ANALYSIS. Available: http://www.greenpasture.org/utility/showDocumentFile/?objectID=88. Last accessed 8/11/16.
5. Robert G. Ackman (1989). Marine Biogenic Lipids, Fats, and oils. United States: CRC PRESS. Chapter 8.
6. Bover-Cid S. (2001). Amino acid-decarboxylase activity of bacteria isolated from fermented pork sausages. Int J Food Microbiol. 66 (3), 185-9.
7. Decker, E. A. and Xu, Z. 1998. Minimising rancidity in muscle foods. Food Technol. 52 (10), 54-59.
8. Kartha VN, Krishnamurthy S.. (1977). Antioxidant function of vitamin A. Int J Vitam Nutr Res. 47 (4), 394-401.
9. Antonio Ayala. (2014). Lipid Peroxidation: Production, Metabolism, and Signaling Mechanisms of Malondialdehyde and 4-Hydroxy-2-Nonenal. Oxidative Medicine and Cellular Longevity. 2014 (1), 0-31.
Images courtesy of Michael Jefferies, Ancient Nabeul and Andre Skibinski.

